Research
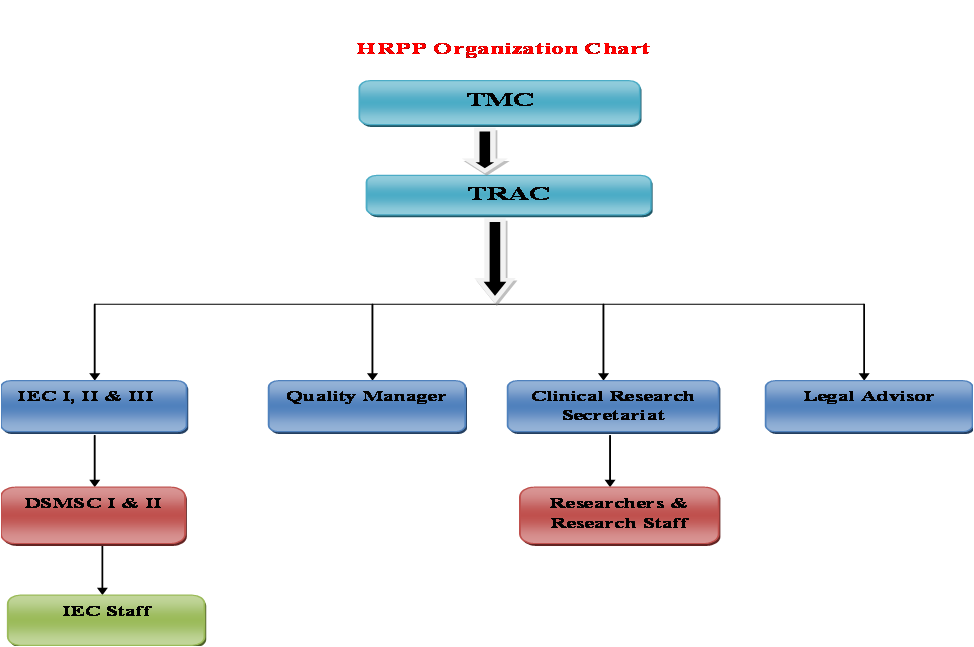
TRAC has been constituted in the year 2008. The main objective of this council is to support and improve the quality of basic, translational and clinical research in TMC with specific focus on the following areas:
- Periodically review the progress of scientific and clinical research
- Set directions, priorities and thrust areas for research
- Suggest and review proposals for collaborations between within TMC and with other National or International Institutions
- Review sponsored research proposals for feasibility
- Coordinate the functioning of committees for research and ethics in TMC
- Review the expenditure and income incurred on hospital services, laboratory and administrative functions for investigator initiated and sponsored research conducted in TMC.
TRAC Membership :
- Chairperson – Director, TMC
- Members – Director, TMH, Director Academics, Director ACTREC, Dy-Director ACTREC,
Medical Superintendent
Member secretary IEC I, IEC II, IEC II, Secretary DSMSC, OIC CRS, Coordinator, TRAC
REGISTRATIONS :
As per gazette notification of May 2013, IRBs were registered with Drug Controller General India (DCGI) as “Institutional Ethics Committee” IEC-I, IEC-II and IEC-III.
- IEC-I : ECR/170/Inst/MH/2013 issued under Rule 122DD of the Drugs & Cosmetic Rules 1945
- IEC-II: ECR/414/Inst/MH/2013 issued under Rule 122DD of the Drugs & Cosmetic Rules 1945.
- IEC-II: ECR/149/Inst/MH/2013 issued under Rule 122DD of the Drugs & Cosmetic Rules 1945.
Department of Health and Human Services (DHHS) through the Office for Human Research Protections (OHRP) Registrations
Institution has a Federal Wide Assurance (FWA) number - FWA00006143
IEC-I IORG No. IRB00003414
IEC-II IORG No. IRB00007802
IEC-III IORG No. IRB00009642
The committee regular meets to discuss different policies pertaining to clinical research , infrastructure and intramural funding.
प्रशंसा पत्र
डीएमजी
Convener : - Dr. Navin Khattry
Secretary : - Dr. Sumeet Gujral
Convener : Dr. Bharat Rekhi
Secretary : Dr. Ashish Gulia
Convener : Dr. Tanuja Shet
Secretary : Dr. Ashwini Budrukkar
Convener : Dr. Mukta Ramadwar
Secretary : Dr. Reena Engineer
Convener: Dr. Prathamesh Pai
Secretary: Dr. Kumar Prabhash
Convener: Dr. Prathamesh Pai
Secretary: Dr. Kumar Prabhash
Convenor: Dr. Tejpal Gupta
Secretary: Dr. Epari Sridhar
Convener : Dr. (Surg Cdr) Gaurav Narula
Secretary : Dr. P. G. Subramanian
Convener : Dr. Seema Kembhavi
Secretary : Dr. Tushar Vora
Convener : Dr. Sarbani Ghosh Laskar
Secretary : Dr. Nilendu Purandare
Convener : Dr. Ganesh Bakshi
Secretary : Dr. Amit Joshi
शिक्षा
ग्लोबल नेविगेशन
हमसे संपर्क करें
टाटा स्मारक अस्पताल
डॉ ई बोर्जेस रोड, परेल, मुंबई - 400 012 भारत
फ़ोन: +91-22- 24177000, 24146750 - 55
फैक्स: +91-22-24146937
ईमेल : msoffice@tmc.gov.in (रोगी देखभाल और प्रश्नों के लिए) / hrd@tmc.gov.in(प्रशासनिक के लिए - HRD मायने रखता है)